Understanding The Role Of Nitrogen In Chemical Reactions
In the realm of chemistry, nitrogen plays an essential role in various reactions, particularly in the formation of compounds vital for life. The significance of nitrogen is often understated, yet its presence can dramatically alter the outcome of a reaction. To fully grasp the impact of nitrogen on chemical processes, one may ponder: how can we conclude that the reaction started with three nitrogen atoms? This inquiry opens the door to a deeper understanding of molecular interactions and the foundational role nitrogen plays in the synthesis of different substances.
As we delve into the world of chemical reactions, it becomes clear that nitrogen's unique properties allow it to participate in diverse ways. The molecular structure of nitrogen, particularly in its diatomic form, shows its potential to bond with various elements, leading to the formation of critical compounds such as ammonia and nitrates. When we examine specific reactions involving nitrogen, it is crucial to consider the initial state of reactants and how they contribute to the overall process.
Moreover, the question of whether we can conclusively determine that a reaction began with three nitrogen atoms is not merely academic; it has real-world implications. Understanding the starting materials of a reaction allows chemists to predict outcomes, optimize processes, and develop new materials with desirable properties. As we explore this topic, we will address some fundamental questions surrounding the role of nitrogen and how it influences various chemical reactions.
What is the Significance of Nitrogen in Chemistry?
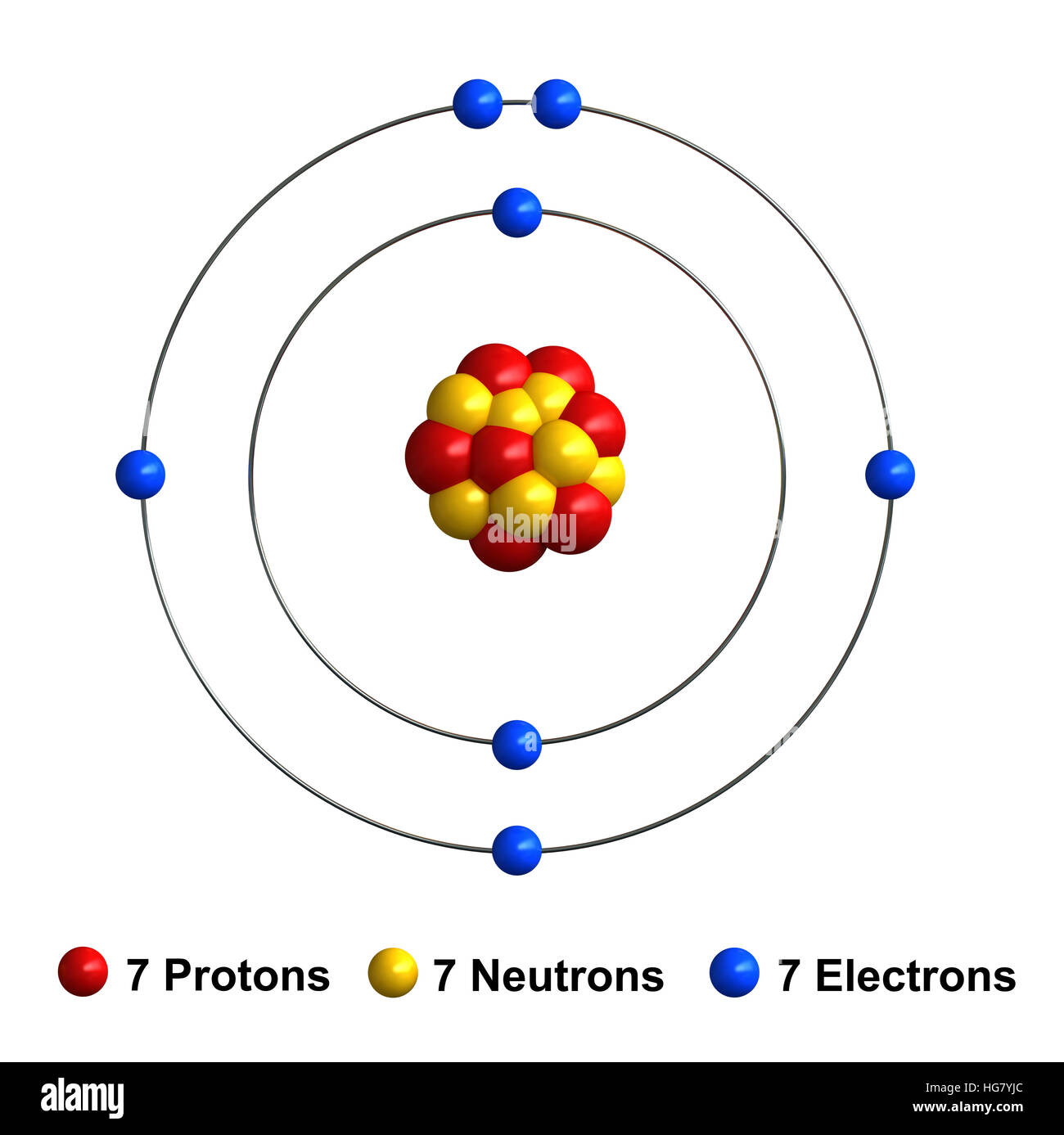
Nitrogen is a vital element found in the atmosphere and is a fundamental building block in many biological and chemical processes. Here are some key points about its significance:
- Nitrogen makes up approximately 78% of the Earth's atmosphere.
- It is an essential component of amino acids, proteins, and nucleic acids.
- Nitrogen is crucial in agriculture as it is a primary nutrient in fertilizers.
- It plays a role in various chemical reactions, including the synthesis of explosives and pharmaceuticals.
How Do We Identify the Reactants in a Chemical Reaction?
Identifying the initial reactants in a chemical reaction is vital for understanding the overall process. Here are some common methods:
- Stoichiometry: This involves using the balanced chemical equation to determine the quantities of reactants and products involved.
- Spectroscopy: Techniques such as infrared and nuclear magnetic resonance spectroscopy can help identify molecular structures and their components.
- Kinetic Studies: By observing the rate of reaction and changes in concentration over time, chemists can infer the initial reactants.
Can We Conclude That the Reaction Started with Three Nitrogen Atoms?
To answer this question, we must first look at the specific chemical reaction in question. For example, consider the formation of ammonia (NH₃) from nitrogen (N₂) and hydrogen (H₂). The balanced equation is:
N₂ + 3H₂ → 2NH₃
In this reaction, we see that one molecule of nitrogen reacts with three molecules of hydrogen to produce two molecules of ammonia. While we cannot conclude that the reaction started with three nitrogen atoms, we can examine the role of nitrogen within the context of the reaction.
What Other Reactions Involve Nitrogen Atoms?
Nitrogen is involved in numerous reactions beyond the formation of ammonia. Here are a few examples:
- Nitric Acid Production: The Ostwald process involves the oxidation of ammonia to form nitric acid.
- Nitrogen Fixation: Certain bacteria convert atmospheric nitrogen into ammonia, a process critical for soil fertility.
- Synthesis of Urea: The Haber-Bosch process combines nitrogen and hydrogen to produce ammonia, which is then converted into urea.
What Are the Implications of Nitrogen in Environmental Chemistry?
Nitrogen compounds have significant implications for environmental chemistry. Here are some areas of concern:
- Eutrophication: Excessive nitrogen from fertilizers can lead to nutrient pollution in water bodies, causing harmful algal blooms.
- Greenhouse Gas Emissions: Nitrous oxide (N₂O), a nitrogen compound, is a potent greenhouse gas contributing to climate change.
- Acid Rain: Nitrogen oxides can lead to the formation of acid rain, impacting ecosystems and human health.
How Can We Study the Role of Nitrogen in Reactions?
Studying the role of nitrogen in chemical reactions can be approached through various methods:
- Experimental Analysis: Conducting laboratory experiments to observe the outcomes of reactions involving nitrogen.
- Theoretical Modeling: Using computational chemistry to predict reaction pathways and product formation.
- Field Studies: Observing nitrogen's role in natural processes, such as nitrogen fixation in soil.
What Are the Future Directions for Nitrogen Research?
The future of nitrogen research is promising, with several exciting directions to explore:
- Green Chemistry: Developing sustainable methods for nitrogen fixation and reducing environmental impact.
- Biotechnology: Utilizing nitrogen-fixing bacteria for agricultural advancements.
- Climate Change Mitigation: Investigating ways to reduce nitrogen oxide emissions and their impact on global warming.
Can We Conclude That the Reaction Started with Three Nitrogen Atoms?
In conclusion, while it is not accurate to state that a reaction started with three nitrogen atoms in every case, we can affirm that nitrogen's presence is crucial in many chemical reactions. Through careful examination and analysis, we can understand how nitrogen atoms participate and contribute to the formation of products. This understanding is essential for advancing both theoretical knowledge and practical applications in chemistry.
Final Thoughts on Nitrogen's Role in Chemistry
As we have explored, nitrogen is a fundamental element in various chemical reactions, influencing the outcome and properties of numerous compounds. By asking questions such as, "Can we conclude that the reaction started with three nitrogen atoms?" we engage in a deeper understanding of the intricate relationships between elements in chemical processes. The future of nitrogen research holds great potential, and continued exploration will undoubtedly yield new insights and advancements in the field.


ncG1vNJzZmixn6PAtr7IZqWeq6RjsLC5jq2pnqaUnruogY6cpqebnKqxpnnToZitZaSdsm6%2BxJqaraGfo3q0wMCrq56cXay2tbSMrZ%2BrnZViu6rA0aienqZdlsGwudJnn62lnA%3D%3D